“Viagra for the brain.” It’s a slogan designed to attract the attention of consumers concerned about cognition. Then there was a massive online ad campaign of “news” websites featuring supposed testimonials from people like Bill Gates and the now-late Dr. Stephen Hawking. It’s no wonder people forked over millions for supplements that went by names like Geniux, Xcel, EVO, and Ion-Z. But according to proposed FTC settlements, the entire promotion was riddled with deception. If you have clients in the affiliate marketing ecosystem, the case merits particular attention.
The defendants marketed the Geniux products on their own websites as “smart drugs” that would – among other things – improve short- and long-term memory even in people experiencing cognitive decline, boost focus and concentration, prevent memory loss, and increase IQ. The defendants also claimed that “clinical trials have shown that Geniux boosts brain power by up to 89.2% [and] increases focus by up to 121%.”
That’s not the only way the defendants pitched their products. They also made extensive use of at least 36 different affiliate networks who, in turn, had affiliate marketers blanketing the internet with ads. (You know how that works. If a consumer views an affiliate marketer’s ad and takes a specific action – for example, clicking through to a site run by the defendants and buying the product – the defendants pays a fee to the affiliate network, who then pays a fee to the affiliate marketer. When the claims at issue are false or misleading, think of it as an echo chamber of deception.)
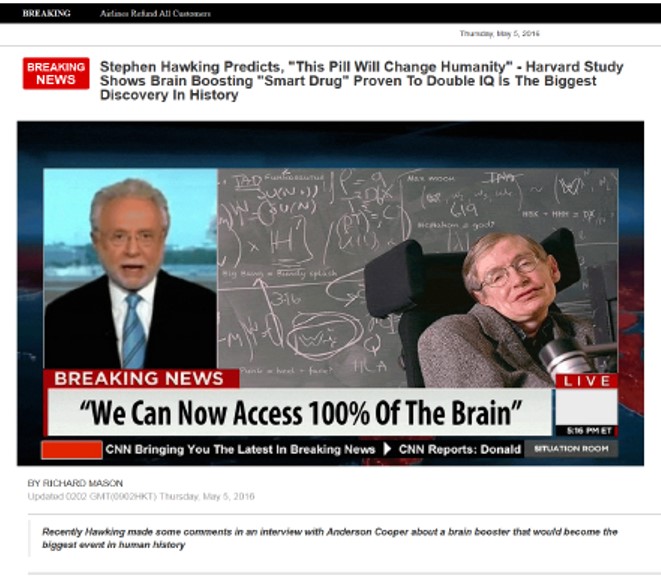 One pervasive marketing technique was sham news sites that used well-known names to tout the products. For example, a purported CNN news story claimed that reporter Anderson Cooper had tried Geniux and concluded it’s “the real deal. The increase in focus, creativity and overall mental performance was a little bit scary to be honest – I felt like a different person.” According to another CNN “report,” “Stephen Hawking said that his brain is sharper than ever, more clear and focused and he credits a large part to using Geniux.” The same piece included a photo of Bill Gates and this quote: “The results were unbelievable. Every aspect of my mental performance accelerated from day 1. A must try.”
One pervasive marketing technique was sham news sites that used well-known names to tout the products. For example, a purported CNN news story claimed that reporter Anderson Cooper had tried Geniux and concluded it’s “the real deal. The increase in focus, creativity and overall mental performance was a little bit scary to be honest – I felt like a different person.” According to another CNN “report,” “Stephen Hawking said that his brain is sharper than ever, more clear and focused and he credits a large part to using Geniux.” The same piece included a photo of Bill Gates and this quote: “The results were unbelievable. Every aspect of my mental performance accelerated from day 1. A must try.”
Some of the “articles” featured comments from consumers who supposedly used the products. For example, “Dan” posted, “After I started taking Geniux I got a promotion at work after just 3 weeks. Three months later I’m CEO and have surpassed all my colleagues.” Given his recent success, it’s doubtful “Dan” wanted his money back, but other consumers were lured in with promises of a “100% MONEY BACK GUARANTEE.”
But the FTC says the promotion was rife with illegality. There wasn’t valid science to back up the cognition promises, the claims about clinicals were false, those “news” sites weren’t news sites at all, none of the big names dropped in the ads had anything to do with the promotions, and even commenters like “Dan” were phony. What’s more, consumers who asked for refunds were often stymied at every turn with contradictory conditions, unanswered customer service phone lines, and “the check is being processed” stall tactics.
Among other things, the proposed orders require the defendants to have randomized clinical testing to support any disease claim and a host of cognition-related representations. They’re also barred from using sham news sites, bogus celebrity or consumer endorsements, and a broad range of other promotional techniques related to the conduct alleged in the complaint. In addition, the orders prohibit deceptive claims about cost, returns, refunds, billing, etc. Other provisions spell out what the defendants must do to keep tabs on what their affiliate networks and affiliate marketers are up to. One order imposes a $14.5 million judgment, which will be partially suspended after the defendants pay $523,000. The second order includes an $11.5 million financial remedy, which will be partially suspended when one defendant pays $100,000. Each judgment will come due if it turns out the defendants misrepresented their financial conditions.
What can other companies take from the case?
Have your science in gear before turning the ignition on cognition. Boomer consumers are concerned about cognitive decline, so advertisers are rushing in with preventatives and cures, often lacking in proof. That’s why misleading claims about memory and brain function remain a key FTC priority.
Bogus testimonials and phony formats will land marketers in legal hot water. Whether it’s a false claim that a famous person uses a product or a glowing recommendation from a non-existent consumer, deceptive endorsements violate the FTC Act. The same holds true for ads that mislead consumers into thinking they’re news sites or other kinds of independent content.
Twisted paths won’t throw legal bloodhounds off the scent. The complexity of the defendants’ corporate structures was rivaled only by its labyrinth of affiliate relationships. Convoluted networks don’t deter law enforcers and they won’t shield appropriate parties from liability. Look no further than the complaint in this case, which names merchants, an advertising service company, fulfillment companies, a payment processor, holding companies, and four individuals.
Monitor what others are doing to help sell your product. “We don’t know who our affiliates are or what they’re up to.” It’s a common refrain, but it doesn’t cut much ice when consumers have been deceived. The orders in this case require the defendants to take specific steps to change their head-in-the-sand approach to affiliate marketing.
